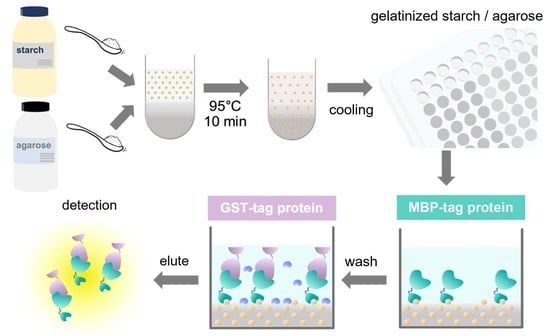
MPs | Free Full-Text | A Cost-Effective Immobilization Method for MBP Fusion Proteins on Microtiter Plates Using a Gelatinized Starch–Agarose Mixture and Its Application for Convenient Protein–Protein Interaction Analysis
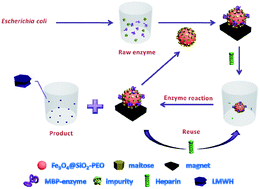
Magnetic nanoparticles for the affinity adsorption of maltose binding protein (MBP) fusion enzymes - Journal of Materials Chemistry (RSC Publishing)
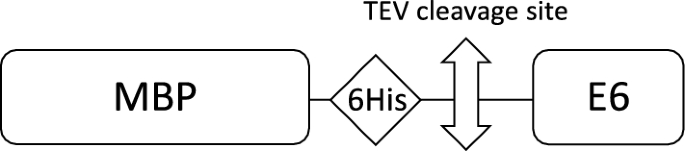
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text

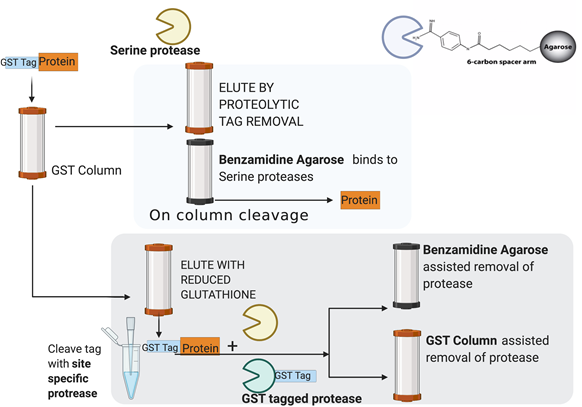
Two-step metal affinity purification of double-tagged (NusA–His6) fusion proteins | Nature Protocols

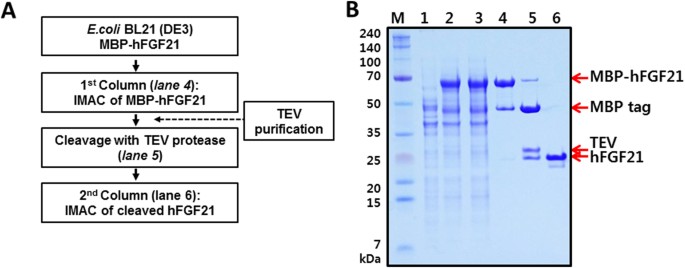
High-yield soluble expression, purification and characterization of human steroidogenic acute regulatory protein (StAR) fused to a cleavable Maltose-Binding Protein (MBP) - ScienceDirect
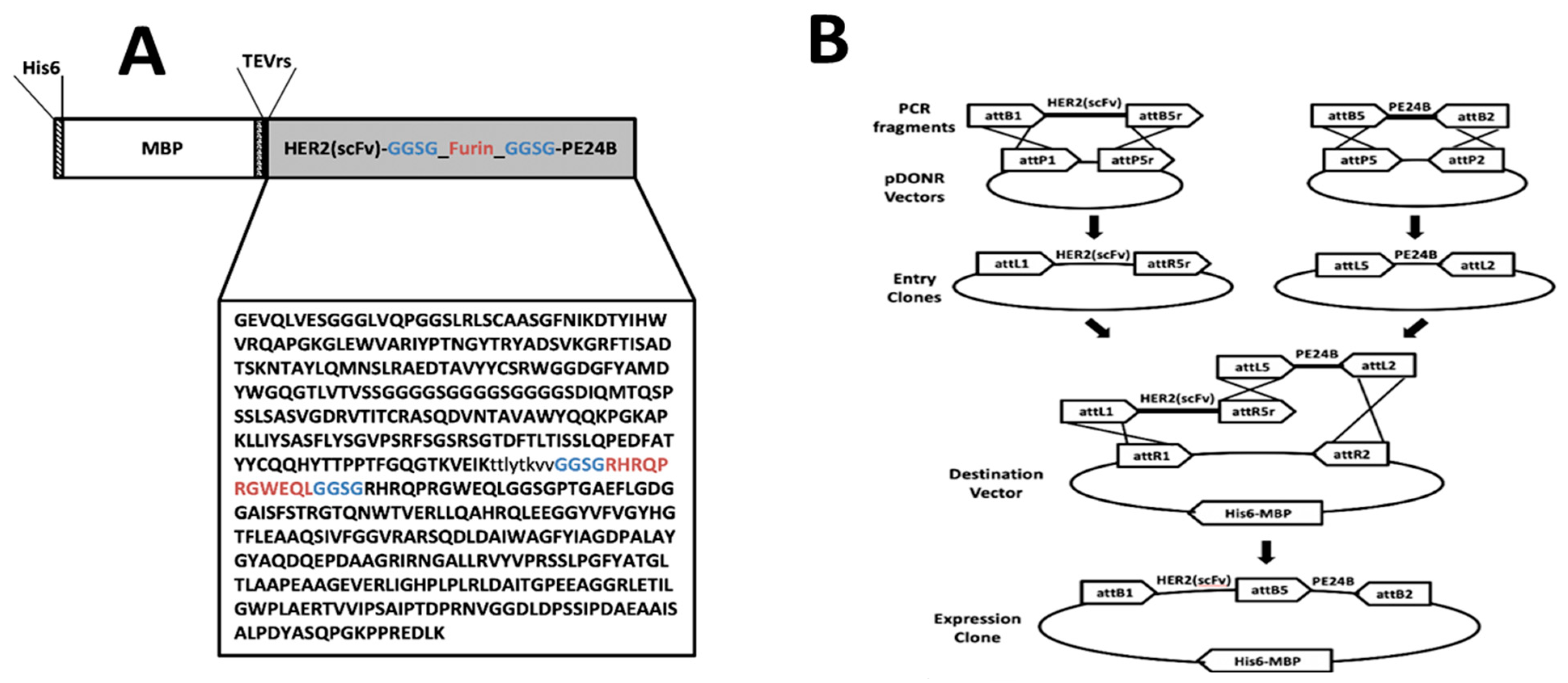
IJMS | Free Full-Text | Soluble Cytoplasmic Expression and Purification of Immunotoxin HER2(scFv)-PE24B as a Maltose Binding Protein Fusion

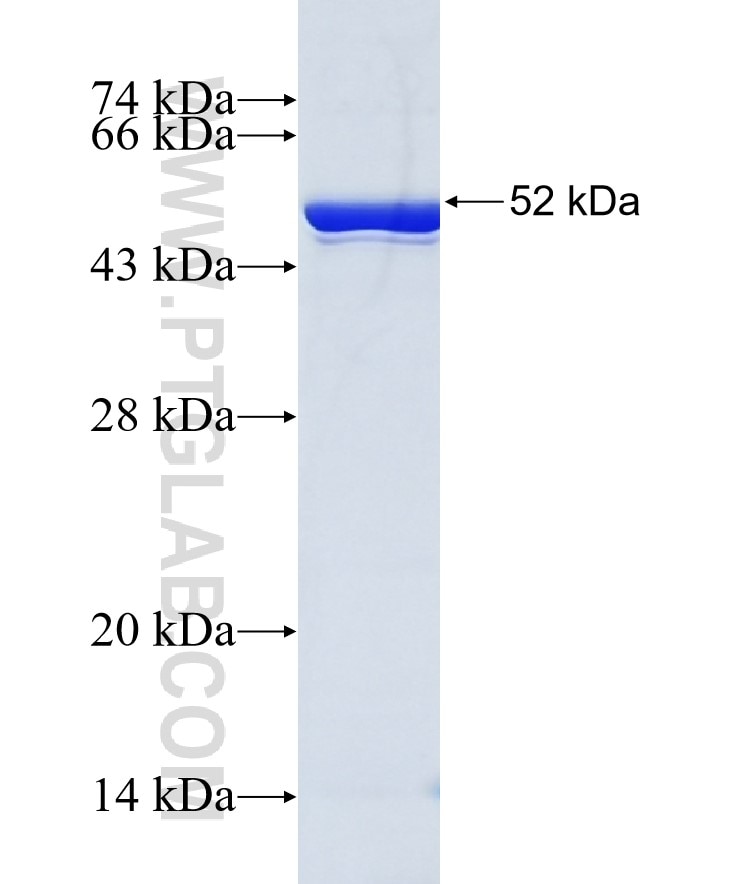
Systematic analysis of the expression, solubility and purification of a passenger protein in fusion with different tags - ScienceDirect
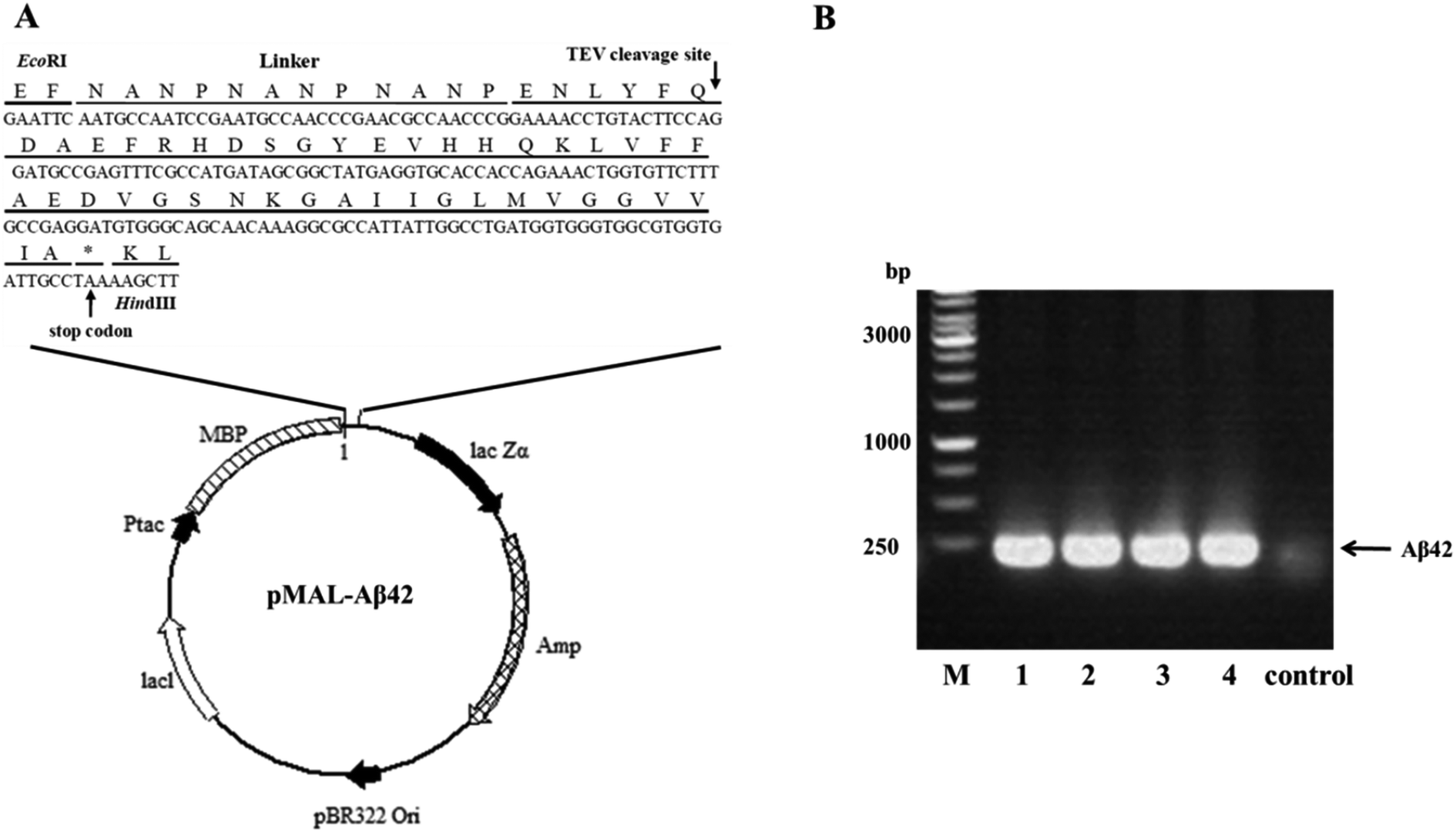
Highly efficient soluble expression, purification and characterization of recombinant Aβ42 from Escherichia coli - RSC Advances (RSC Publishing) DOI:10.1039/C8RA00042E

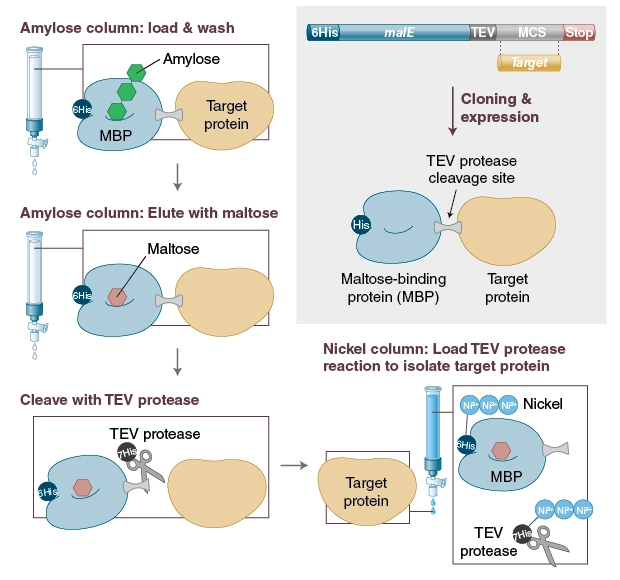
Affinity Purification of a Recombinant Protein Expressed as a Fusion with the Maltose-Binding Protein (MBP) Tag. | Semantic Scholar